Miscible Homopolymer Blends
In rare instances, two homopolymers will be miscible, exhibiting lower critical solution temperature (LCST) behavior. Such polymer pairs form a homogeneous blend at room temperature and phase separate at higher temperatures. The origin of miscible phase behavior is an attractive interaction between the monomeric species of the two homopolymers, described by the Flory-Huggins interaction parameter, χ. Fundamentally, this can happen when two monomer species are chemically similar, like polyisobutylene/polybutadiene (PIB/PBD), or shaped in such a way as to enable dispersion interactions (polystyrene/polyvinyl methyl ether). My own work has measured χ for a series of PIB/dPBD blends (dPBD is deuterated for measurement contrast), revealing a rarely observed dependence on both the composition and molecular weight.
Blends at the critical composition transition directly from the homogenous to the two-phase state without passing through a metastable window. Critical blends are ideal for phase characterization studies because they do not risk kinetic trapping, although we found that even off-critical blends equilibrated rapidly because both homopolymers are rubbery at room temperature. Small-angle neutron scattering (SANS) was selected to characterize the phase of each blend during heating ramps because it captures the length scales immediately preceding critical fluctuations. Extrapolations of the inverse structure factor in the limit of zero scattering angle (called the susceptibility) measured at temperatures near the critical point can be used to pinpoint the spinodal temperature (for off-critical blends) or the critical temperature (for critical blends). Measuring the binodal temperature in off-critical blends is more experimentally challenging because the observed temperature of phase separation may be affected by the kinetics of nucleation and growth of the two phases.
Thermodynamic modeling and experiment using Flory-Huggins Theory
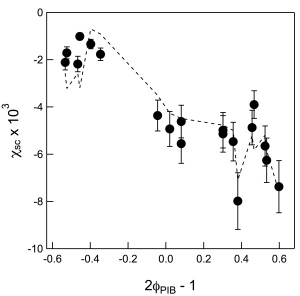
The temperature-dependent Flory-Huggins χ was measured by fitting the random phase approximation (RPA) to SANS spectra from different blends at different temperatures1. Although the strict definition of χ requires independence from composition and chain length, this definition depends on the validity of a lattice-based approached. Miscible polymer blends often exhibit off-lattice effects, or apparent compressibility, similar to the net volume reduction upon mixing sugar into water. This directly leads to an observed composition-dependent χ. Molecular weight has a lesser effect on χ, but presumably the effect of chain ends is more significant in off-lattice systems. We found that the data could be well described by an empirical form of χ that incorporated a direct dependence on composition and inverse dependence on the (geometrical) average molecular weight of the two homopolymers. Fig. 1 shows measured values of χ for PIB/dPBD along with the empirical fit.

The Flory-Huggins theory predicts the phase behavior of polymer blends for which χ (along with the molecular weight of each species and composition of the blend) is known. However, because the measured form of χ is composition-dependent, it cannot be directly used to predict phase behavior. Rather, we must distinguish the value measured from scattering, χsc, from the value used for thermodynamic predictions, χFH. Making this correction leads to astonishingly accurate phase predictions across a series of PIB/dPBD blends2. As observed with other LCST systems, the binodal and spinodal both tend to flatten in the vicinity of the critical point, more so for blends in which the polymers have dissimilar molecular weights. The asymmetry of the phase diagram itself, is relatively mild, and slightly more pronounced as the polymer molecular weights become more similar.
